Chemists discovered that a seemingly solid, nonporous crystal can 'come alive' when heated. A two-step transformation releases trapped molecules, drives a vivid blue - green - yellow glow, and even propels the crystal forward through bubble-powered motion.(Nanowerk News) In a study recently published in Angewandte Chemie International Edition ("Blue–Green–Yellow Thermochromic Fluorescence and Bubble‐Propelled Crystal Motion Driven by Molecular Gear Rotations"), researchers from National Taiwan University report that a seemingly solid, nonporous organic crystal can undergo dramatic structural and mechanical transformations when gently heated. |
|
Although such crystals appear tightly packed and impermeable, the team discovered that heating can trigger a precisely orchestrated, two-step internal rearrangement that releases trapped solvent molecules while producing striking optical and mechanical responses. |
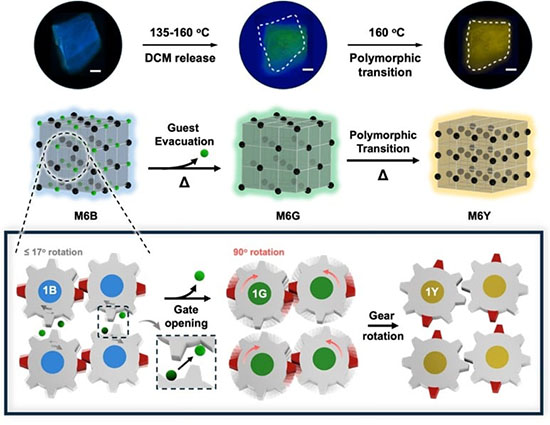
| The material, a pentiptycene-derived organic solvate crystal, initially emits blue light. Upon heating, it undergoes a first crystal-to-crystal transformation in which subtle, gear-like molecular rotations transiently create zigzag diffusion pathways inside the lattice. |
| These temporary channels allow dichloromethane (DCM) molecules to escape, shifting the crystal’s fluorescence from blue to green. When the process occurs in silicone oil, the escaping solvent forms microscopic bubbles asymmetrically on the crystal surface. The resulting bubble evolution generates directional propulsion, causing the crystal to move visibly under a microscope. |
| A second heating step induces a larger, highly correlated molecular rotation of approximately 90 degrees. This rearrangement alters the packing of anthracene units and the conformation of alkyl chains, producing a new yellow-emissive phase dominated by excimer formation. The overall process results in a vivid blue → green → yellow fluorescence sequence coupled with guest release and autonomous motion. |
| “These crystals may look static, but at the molecular level they behave like tiny machines,” said corresponding author Prof. Jye-Shane Yang. |
| “What surprised us most was that a nonporous crystal can temporarily reorganize itself to let molecules escape, and even convert that process into visible motion. This discovery opens new possibilities for designing responsive solid materials that integrate structure, light emission, and mechanics in a programmable way.” |
| Source: National Taiwan University (Note: Content may be edited for style and length) |