A mitochondrial enzyme, Ak4, strengthens immune defense by promoting mitochondrial DNA synthesis and enhancing macrophage antibacterial activity.
When bacteria invade the body, macrophages serve as frontline defenders, rapidly eliminating pathogens to protect tissue integrity. One of their key weapons is mitochondrial reactive oxygen species (mtROS). However, how macrophages enhance this antibacterial function has remained poorly understood.
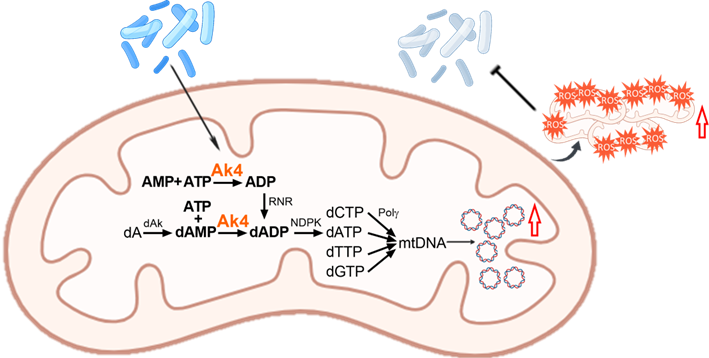
In a study published in the Journal of Experimental Medicine (April 6, 2026; 223(4): e20250978), researchers from National Taiwan University uncovered a metabolic mechanism that strengthens macrophage defense. They found that bacterial infection triggers an increase in mitochondrial DNA (mtDNA), which in turn boosts mtROS production and promotes efficient bacterial clearance.
At the center of this process is adenylate kinase 4 (Ak4), a mitochondrial enzyme that is strongly induced during infection. Loss of Ak4 impairs mtDNA synthesis, reduces mtROS production, and weakens macrophage antibacterial activity. Mice lacking Ak4 exhibit higher bacterial burdens and increased susceptibility to infection.
Mechanistically, Ak4 regulates nucleotide metabolism required for mtDNA replication. Disruption of its enzymatic activity abolishes this effect, demonstrating that Ak4 directly links cellular metabolism to immune defense.
These findings reveal a previously unrecognized pathway connecting mitochondrial function and innate immunity, suggesting new opportunities to modulate immune responses in infectious and inflammatory diseases.
“Ak4 acts as a key metabolic regulator that enables macrophages to mount effective antibacterial responses,” says corresponding author Prof. Shi-Chuen Miaw at Graduate Institute of Immunology, National Taiwan University.
Prof. Shi-Chuen Miaw's email address: smiaw@ntu.edu.tw