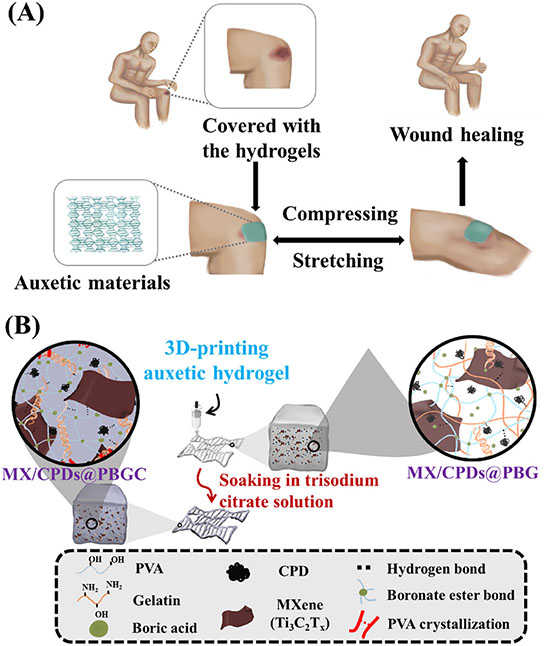
A 3D-printed hydrogel wound dressing loaded with MXene and carbonized polymer dots kills drug-resistant MRSA and speeds healing in diabetic mice.(Nanowerk News) Researchers have developed a 3D-printed hydrogel wound dressing infused with two advanced nanomaterials to treat chronic wounds infected by multidrug-resistant bacteria. The dressing, designated MX/CPDs@PBGC, integrates Ti3C2Tx MXene nanosheets and carbonized polymer dots into a self-healing auxetic polymer matrix, and in animal testing it outperformed a commercial antibacterial bandage in healing diabetic wounds colonized by MRSA. The study was published in Small ("3D‐Printed Auxetic Hydrogel Dressings Reinforced With MXene and Carbonized Polymer Dots for Cascaded Antibacterial and Pro‐Regenerative Effects in Wound Healing"). |
Key Findings
|
| Antimicrobial resistance poses a growing threat to wound care, particularly for patients with chronic conditions such as diabetes. Wounds in diabetic patients are often colonized by bacteria like methicillin-resistant Staphylococcus aureus (MRSA), which resist standard antibiotic treatments. Conventional wound dressings typically provide a passive physical barrier but lack the capacity to actively eliminate resistant pathogens or adapt to body movement during wear. |
|
The therapeutic core of the new dressing is a pair of nanomaterials embedded in its hydrogel matrix. Ti3C2Tx MXene nanosheets contribute antioxidant and anti-inflammatory activity by neutralizing free radicals that typically impede wound repair. Carbonized polymer dots (CPDs), synthesized from mannose and arginine, act as the primary antibacterial agents. Rather than relying on conventional antibiotics, these nanoparticles physically damage bacterial cell membranes, destroying drug-resistant organisms through direct mechanical action. |
| The dressing also addresses the mechanical shortcomings of existing wound products. Its auxetic geometry causes the material to expand laterally when stretched rather than thinning as conventional materials do. This property enables it to maintain consistent contact with wound surfaces on high-movement body areas such as knees and elbows without peeling or losing its seal. |
| The base matrix consists of polyvinyl alcohol, boric acid, gelatin, and citrate (PBGC). Dynamic bonds within this network give the hydrogel self-healing capability, allowing it to restore its internal structure after mechanical stress or minor tearing. This ensures the dressing maintains a continuous protective barrier even during physical activity. |
| The hydrogel's release behavior is tuned to respond to environmental conditions at the wound site. At room temperature the dressing remains stable, but at body temperature (37°C) it degrades and releases its therapeutic cargo more rapidly. It also reacts to the acidic pH characteristic of infected wounds, concentrating its antibacterial payload where infection is most active. This dual-responsive release mechanism directs treatment to the point of need while reducing waste of active agents. |
| Testing in a diabetic mouse wound model infected with MRSA showed that the MX/CPDs@PBGC dressing outperformed a commercial 3M antibacterial bandage across multiple healing metrics. Beyond direct bacterial killing, the hydrogel influenced the immune response by shifting white blood cells from a pro-inflammatory state toward a regenerative phenotype. This immune reprogramming translated into faster wound closure, greater collagen deposition, and enhanced formation of new blood vessels in treated animals. |
| "By combining the structural adaptability of 3D printing with the molecular precision of nanotechnology, this hydrogel offers a comprehensive strategy for managing the most stubborn wounds and represents a significant leap forward in the field of regenerative medicine," said co-corresponding author Prof. Tzu-En Lin from the Institute of Applied Mechanics at National Taiwan University. |
| By uniting nanomaterial-driven therapy, mechanical adaptability, and environment-responsive drug delivery in a single 3D-printed platform, the dressing addresses several persistent challenges in chronic wound management simultaneously. If the approach translates successfully from animal models to clinical use, it could expand treatment options for diabetic patients with wound infections that no longer respond to standard antibiotics. |
To see article on nanowerk:
https://www.nanowerk.com/nanotechnology-news3/newsid=68940.php